|
Similarly, the isotope whose atomic weight is 24.986 amu has a mass number of 25, 13 neutrons, and 25 Mg as a symbol. 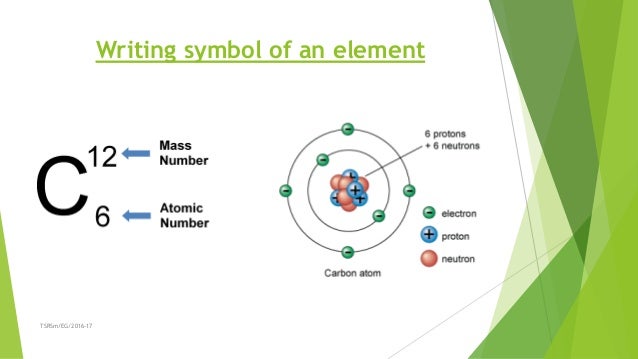
The isotope whose atomic weight is 23.985 u has a mass number of 24 (protons and neutrons), so 24 - 12 protons gives 12 neutrons. Learners read definitions of atomic symbols, atomic numbers, and mass numbers and then answer questions about the number of neutrons, protons, and electrons. You will need to refer to a periodic table for proton values. There are 12 protons in all magnesium isotopes. In this notation, the atomic number is not included. The first letter of symbol is always written as. Symbol-mass format for the above atom would be written as Cr-52. Berzilius suggested that the symbols of elements be made from one or two letters of the name of the element. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. This atomic number is ordinarily given the symbol Z. An atom is first identified and labeled according to the number of protons in its nucleus. An isotope is one of two or more species of atoms of the same chemical element that have different atomic mass numbers (protons + neutrons). Every chemical element has one or more isotopes. Since 1961 the standard unit of atomic mass has been one-twelfth the mass of an atom of the isotope carbon-12. The "A" value is written as a superscript while the "Z" value is written as a subscript. atomic weight, also called relative atomic mass, ratio of the average mass of a chemical element’s atoms to some standard. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
